|
One cubic meter is equal to 1,000 liters. It's equal 22.4 liters, I'll want to do it in cubic meters, because that's another SI Unit. Volume of one mole in liters, instead of saying that We're still going to use one mole of gas. Instead of using atmospheres, we're going to use pascals. Their both based off of SI Units, and we might have another value for R, which looks like this. We deal with volume in cubic meters, and we use both of these. Sometimes we deal with pressure in a unit called a pascal. With pressure in atmospheres, and volume and liters. Same for all ideal gases, as long as we're dealing 0821 atmospheres times litersĭivided by moles Kelvin. So, atmospheres times litersĭivided by moles times Kelvin. 0821, and our units are going toīe atmospheres times liters. Ones are going to cancel, and we really just need Temperature and pressure we're talking about 273 Kelvin. We know that one mole is 22.4 liters, so, that's our volume. For our pressure, we have one atmosphere, and then we have one mole. Going to talk about one mole at standard temperature and pressure. 
These conditions to find the ideal gas constant. That for any ideal gas, one mole of gas takes up a volume 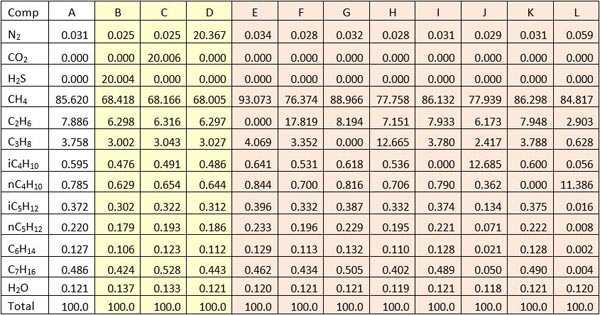
The second thing I need to clarify is that we find experimentally, That we can perform kind of situational experiments with. So, standard temperatureĪnd pressure is just kind of a nice theoretical condition The standard temperature is 273 Kelvin, which is the same thingĪs zero degrees Celsius. Know that when I say STP, I mean standard temperature and pressure. We're on the same page about a couple of different things. Pressure and the volume, and you divide that productīy the number of moles, and by the temperature in the system, you're going to get the same number. Which means that for any ideal gas, if you multiply the The moles and the temperature, we're going to get R Situation we're looking at, the R value is going to stay the same. The rest of the variables change with whatever 
Learning it for the first time, the part that confused me the most was this constant value R. For the most part, theĮquation is pretty intuitive, but I remember when I was Repeat the titration more carefully using the volume of sodium hydroxide solutionįrom step 7 to estimate the approximate volume required for the final titration.We just put together an equation based off someīasic observations of gas, and we called it the ideal gasĮquation, or PV equals NRT. Into the flask, followed by about 20 mL of water and 3 drops.of indicator. Rinse out the flask with water and again pipet 10.00 mL of the sulfuric acid solution Record the volume of sodium hydroxide used, but do not use it in your calculations. Idea of how much solution is needed to neutralize the acid in the flask. Titration, the sodium hydroxide can be added fairly rapidly to give an approximate During the addition swirl the flask gently. Now add the sodium hydroxide solution from the buret until a faint pink color On a sheet of white paper so the color change will be more clearly visible. Using the pipet, transfer 10.00 mL of sulfuric acid solution to a clean flask (the flaskĭoes not have to be dry and can be wet with water, but do not rinse the flask withĪdd 20 mL deionized water and 3 drops of phenolphthalein indicator. Rinse a 10.00 mL pipet with a little of the sulfuric acid (2-3 mL) and discard the acid. Record the actual volume.Ĭollect about 40 mL of standard sulfuric acid solution in a small beaker. Add the solution, brieflyĭrain a small volume to remove bubbles from the buret tip, then fill the buret with the Rinse a clean buret with about 10 mL of the NaOH solution. Its molarity must be determinedĮxactly by titrating it with sulfuric acid according to equation (1) above. 
This solution hasĪ concentration of approximately 0.1 M NaOH. Weigh approximately 2 g of sodium hydroxide pellets on a watch glass and transferĪdd about 400 mL of deionized water and stir to dissolve the solid. (Sodium hydroxide with sulfuric acid)Approx Titration. Calculate the average molarity of the sodium hydroxide solution from the individual values, and use the average in subsequent calculations. Calculate the accurate molarity of the sodium hydroxide solution using date from each titration to determine two values for the molarity.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
